 /GettyImages-1096547948-35b3799817ca4b2fa06888893ef4a348.jpg) The electrons that drop from the higher energy levels to the lower energy levels in an atom release a photon with a specific wavelength, which generates the atomic emission spectrum. Purple is released by highly excited electrons that move long distances. Explain how the atomic emission spectra occur and how they relate to the elements on the periodic table. Because it is no longer at a high energy level, it will emit that same energy in the form of light at that specific wavelength.Įach slit represents one or more electrons that have released energy. The energy used to excite the atom causes the electron to move down a few orbitals, closer to the nucleus. Atomic spectra consist of narrow peaks (a few nanometers bandwidth) providing two types of analytical information: qualitative analysis (characteristic frequency or wavelength of the absorbed or emitted photon) and quantitative analysis (signal intensity at a given wavelength). Physicist Niels Bohr introduced in 1913 his idea of why the atomic spectrum has the characteristics and properties it has. Colors and light split into a spectrum, which scientists can observe using a spectroscope. 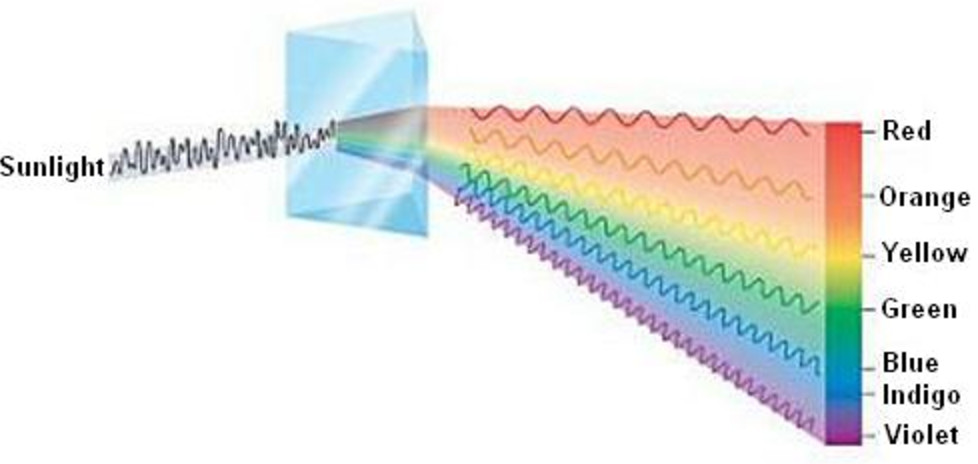 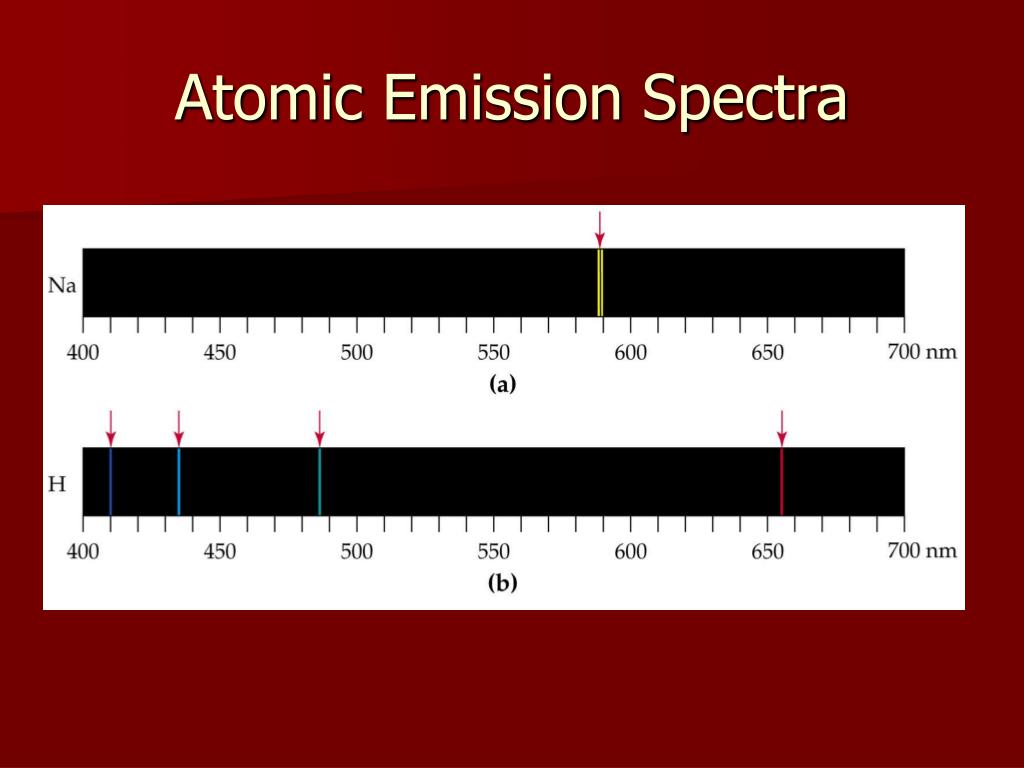
Spectra may be classified according to the nature of their origini.e., emission or absorption. An instrument designed for visual observation of spectra is called a spectroscope, and an instrument that photographs or maps spectra is a spectrograph. This means that, when the atoms of the element are excited with electricity (google how a fluorescent lamp works for an explanation of this), they will give off light only in the same wavelength of energy as the energy put in. The Atomic spectra are defined as the Spectrum of frequencies of electromagnetic radiation emitted or absorbed during transitions of Electrons between. The two best known examples of this are white light through a prism and through water drops, which makes a rainbow. spectrum, in physics, the intensity of light as it varies with wavelength or frequency. are determined by the amount of energy put off.Įach element has its own spectrum because each element has its own electron configuration. For example, because hydrogen is the most basic Atom, it has the most basic Spectrum. #EXAMPLE OF ATOMIC SPECTRA SERIES#It's a series of colored lines, except their spacing, clarity, size, etc. The atomic spectrum is a series of colored lines similar to the rainbow that a prism puts out when white light is sent through it. Every element has a unique atomic spectrum, and you can see it through a diffraction grating. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
